Hepatitis B Virus
3. Cell Culture Systems
In contrast to the intracellular replication steps, our knowledge of the early events of HBV infection -particularly cell attachment, receptor recognition, internalisation and membrane fusion- is still rudimentary. In the past, these problems have been partially circumvented by the usage of primary human hepatocytes. However, due to their very limited availability and the problem of donor specific variability, neither a functional HBV receptor has been identified so far, nor are any details of virus entry (e.g. receptor identity, receptor interaction, route of particle internalisation, mechanism of membrane fusion) known.
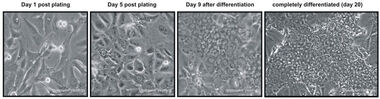
Recently, our close collaborator Philippe Gripon in Rennes described a hepatoma-derived cell line, called HepaRG, that allows experimental induction of differentiation, accompanied by the expression of liver-specific genes (e.g. aldolase B, Cyp 3A4 or Cyp 2E1) (Fig.6) ( Gripon et al, PNAS, 2002 ). In contrast to other hepatoma cells (e.g. HepG2, HuH7), HepaRG cells suffered only minor chromosomal alterations (one trisomy of chromosome 7 and a translocation of chromosome 22 to chromosome 12).
Fig.6: HepaRG cells following different stages of differentiation. : In the undifferentiated state (Fig 1, left), HepaRG cells present a morphology that is comparable to fibroblasts, while 20 days after differentiation they appear hepatocyte-like with visible polarity and the formation of bile ducts (canaliculi).
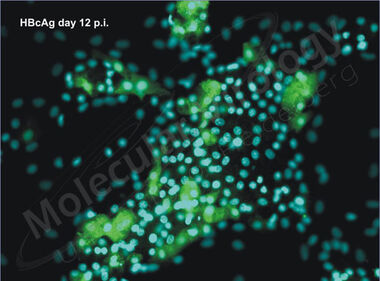
Interestingly, during differentiation HepaRG cells gain susceptibility towards HBV infection (Fig 7). The reason for this phenomenon is not clear hitherto and might be of fundamental interest for our understanding of the HBV infection process.
Fig.7: HBV-core expression in HBV-infected HepaRG cells. Cells were infected with HBV and stained 12 days post infection using an HBV core specific antibody (green). Nuclei are visualized in blue (picture kindly provided by K. Mills).
< Page 2 (Replication) Page 4 (Early Events in Hepadnaviral Infections) >