Lasermikrodissektion
Principle
Laser Microdissection (LMD), also known as Laser Capture Microdissection (LCM), is a contact-free method to isolate subpopulations of tissue cells under direct microscopic visualization. LMD technology can isolate the cells of interest directly or by cutting away unwanted cells to obtain histologically enriched cell populations. A variety of downstream applications exist: DNA analysis, RNA transcript profiling, proteomics and metabolomics. The total time required to carry out this protocol is typically 2 – 8 h and largely depends on the tissue and target-cell type.
Procedure
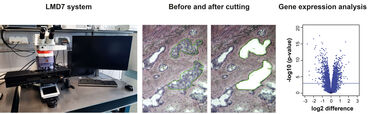
Under a microscope using a software interface, a tissue section (typically 5-50 µm thick) is viewed and individual cells or clusters of cells are identified manually. The laser cutting width is usually less than 1 µm, thus the target cells are not affected by the laser beam. Even live cells are not damaged by the laser cutting and are viable after cutting for cloning and reculturing as appropriate. We are using the LMD7 system by Leica.
Applications
The LMD process does not alter or damage the morphology and chemistry of the sample collected, nor the surrounding cells. For this reason, LMD is a useful method of collecting selected cells for DNA, RNA, protein and/or metabolite analyses. LMD has also been used to isolate acellular structures, such as amyloid plaques. LMD can be performed on a variety of tissue samples including blood smears, cytologic preparations, cell cultures and aliquots of solid tissue. Frozen and paraffin embedded archival tissue may also be used.
For more information please see:
https://en.wikipedia.org/wiki/Laser_capture_microdissection
https://www.leica-microsystems.com/products/light-microscopes/p/leica-lmd7/?country=DE
Equipment:
• Leica LMD7 Laser Capture Microdissection System equipped for sample collection into 96- or 384-well plates
• Agilent 2100 Bioanalyzer
• Cryostat Leica CM1850
• Rotary microtome Microm HM355
Contact:
Publications: