The Füllekrug Lab
Subcellular compartmentalization of lipid metabolism
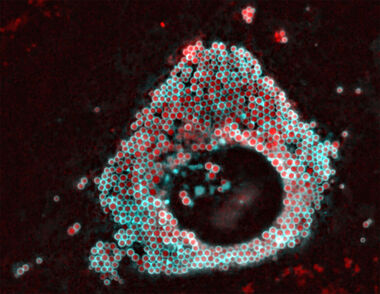
The overall focus of our lab is the intracellular sorting of membrane proteins and lipids, and the functional role of subcellular localization in the trafficking and metabolism of lipids. We are interested in particular how proteins and lipids are interacting to organize the transport and metabolism of fatty acids during the biogenesis of lipid droplets.
Formation of lipid droplets is an essential function of all eukaryotic cells, and the regulation processes involved are highly relevant for the development of obesity and diabetes type II. Biogenesis of lipid droplets starts with the uptake and activation of fatty acids, and we have been characterizing the proteins involved, in particular enzymes of the fatty acyl-CoA synthetase family.
Our model system are mammalian tissue culture cells which are manipulated by techniques of molecular biology, especially through the expression of mutated proteins designed to reveal functional aspects. We combine both microscopy and biochemistry for analysis.
Team
Wissenschaftliche Mitarbeiter/-innen
Wiss. Mitarbeiter/-innen
-
Peiyin Chen, PhD student
Function of perilipin 3 in lipid droplet heterogeneity and metabolism
funded by DFG Research Unit FOR5815 -
Carina Kessler, PhD student
Function of perilipin 3 in lipid droplet heterogeneity and metabolism
funded by DFG Research Unit FOR5815 -

Wentao Mo, M. Sc.
Perilipin-dependent lipid droplet biogenesis and turnover
supported by Chinese Scholarship Council (CSC) -

Wenyue Tian, M.Sc.
Perilipin-3 mediated lipid droplet homeostasis and maturation
supported by the Landesgraduiertenförderung (LGF) Baden-Württemberg
Open positions
We welcome unsolicited applications from students/postdocs interested in the biology of neutral lipid storage. A background in molecular cell biology, bioinformatics, microscopy or fatty acid metabolism is required.
Interested? Please apply by E-Mail, using "Lipid Droplet Researcher" in the subject line and limiting the attachment (one pdf) to 10 MB.
Students of biochemistry (biology, chemistry, biomedicine) are welcome to join the lab for an experimental Master thesis.
All research is relevant for the molecular mechanism of pathogenic processes. The technical approaches comprise molecular biology (cloning, mutagenesis, Cas9 mediated KO and Knock-In), microscopy (immunofluorescence, GFP, confocal analysis), and biochemistry (uptake assays, subcellular fractionation).
Publications
Collaborations
Funding
Landesgraduiertenförderung (LGF) Baden-Württemberg
German Research Foundation (DFG)
Alexander von Humboldt Stiftung
Stiftung Nephrologie Heidelberg
Mobility Programme Heidelberg University









